Fast relief from hives
and itch at week 121
ㅤ
Patient portrayal.
RHAPSIDO® (remibrutinib) demonstrated consistent results in both of its phase 3 trials1
In the REMIX-1 and REMIX-2 trials, RHAPSIDO significantly improved hives, itch, disease activity and angioedema vs placebo in patients with CSU who remained symptomatic on antihistamines. The co-primary end points were absolute change from baseline in HSS7 and ISS7 at week 12.1
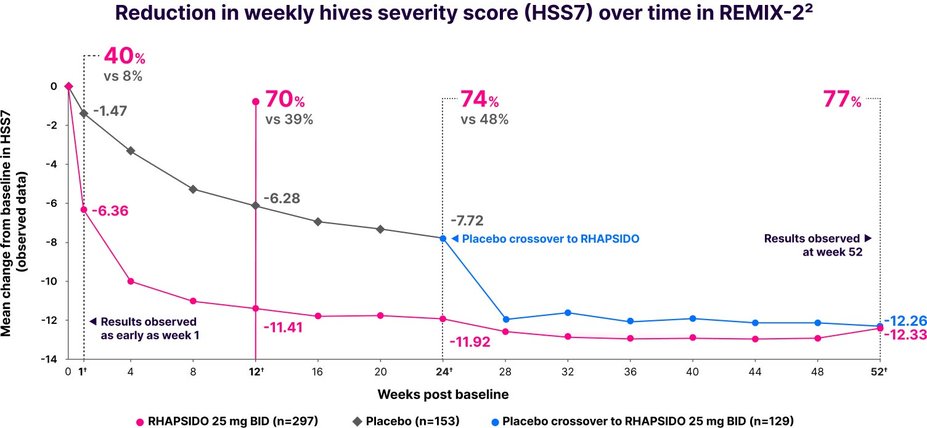
Fast relief from hives1
Significant reduction in HSS7 vs placebo at week 12: LS mean CFB -10.47 with RHAPSIDO vs -6.00 with placebo (P<.001) in REMIX-2. Similar results were observed in REMIX-1.1–3,*
Results were observed as early as week 1 in a post hoc analysis and at week 52 in an exploratory analysis.2,3,†
*Multiple imputation techniques were implemented for missing data.1
†LIMITATIONS: No conclusions or comparisons can be drawn since post hoc or prespecified exploratory analyses were performed at week 1 and weeks 12 and 24, respectively. These analyses were not adjusted for multiplicity. Data after week 24 should be interpreted with caution due to the open-label design and absence of a control group. Week 52 data is a prespecified exploratory analysis and no statistical tests were done.2,4
Improvements in HSS7 at week 12 were consistent regardless of patients' baseline total IgE level.1
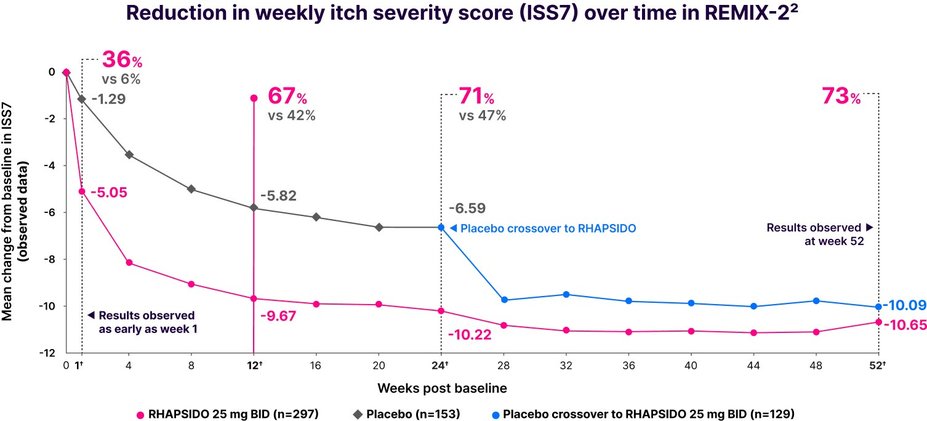
Fast relief from itch1
Significant reduction in ISS7 vs placebo at week 12: LS mean CFB -8.95 with RHAPSIDO vs -5.72 with placebo (P<.001) in REMIX-2. Similar results were seen in REMIX-1.1–3,*
Results were observed as early as week 1 in a post hoc analysis and at week 52 in an exploratory analysis.2,3,†
*Multiple imputation techniques were implemented for missing data.1
†LIMITATIONS: No conclusions or comparisons can be drawn since post hoc or prespecified exploratory analyses were performed at week 1 and weeks 12 and 24, respectively. These analyses were not adjusted for multiplicity. Data after week 24 should be interpreted with caution due to the open-label design and absence of a control group. Week 52 data is a prespecified exploratory analysis and no statistical tests were done.2,4
Improvements in ISS7 at week 12 were consistent regardless of patients' baseline total IgE level.1
RHAPSIDO is proven to deliver well-controlled CSU1,4
Well-controlled CSU (UAS7 ≤6) at weeks 2 and 12 (secondary end points)1,*
*Multiple imputation techniques were implemented for missing data.1
Complete absence of hives and itch (UAS7=0)
At week 12 (secondary end point)1,*
*Multiple imputation techniques were implemented for missing data.1
Exploratory analysis at week 52 (observed data) in REMIX-22,†
†LIMITATIONS: No conclusions or comparisons can be drawn. Data after week 24 should be interpreted with caution due to the open-label design and absence of a control group. Week 52 data is a prespecified exploratory analysis and no statistical tests were done.2
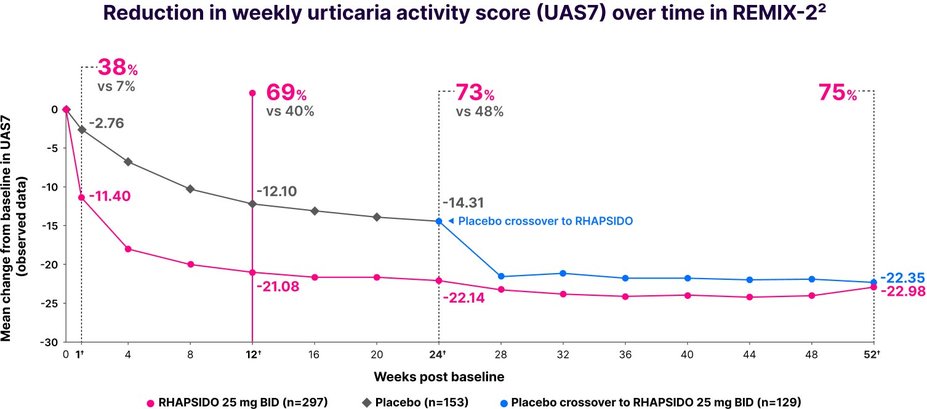
Disease activity results (UAS7)
Key secondary end point: significant reduction in UAS7 vs placebo at week 12 (LS mean CFB in REMIX-2: -19.41 with RHAPSIDO vs -11.73 with placebo; P<.001). Similar results were observed in REMIX-1.1–3,*
*Multiple imputation techniques were implemented for missing data.1
†LIMITATIONS: No conclusions or comparisons can be drawn since post-hoc or prespecified exploratory analyses were performed at week 1 and weeks 12 and 24, respectively. These analyses were not adjusted for multiplicity. Data after week 24 should be interpreted with caution due to the open-label design and absence of a control group. Week 52 data is a prespecified exploratory analysis and no statistical tests were done.2,4
Cumulative number of angioedema-free weeks (AAS7=0) with RHAPSIDO2
In REMIX-2 at baseline:

46% of patients (n=208) had prior angioedema1
Secondary end point LS mean: Cumulative, nonconsecutive, angioedema-free (AAS7=0) weeks vs placebo between baseline and week 12. Similar results were observed in REMIX-1.2,3,*
Cumulative number of angioedema occurrence-free weeks (AAS7=0)
Between baseline and week 12, the LS mean cumulative number of angioedema-free weeks (AAS7=0) was 8.81 with RHAPSIDO vs 6.68 with placebo.2